You must be familiar with GxP referring to the ‘good practice’ guidelines and regulations ensuring that food, medical devices, drugs, and other life science products are safe and of high quality. Good Documentation Practices (GDocP) is one such GxP applicable to the pharmaceutical and medical device industries.
Thoroughly grasping GDocP is paramount for relevant enterprises as it describes a globally recognized standard for the creation, management, and maintenance of reliable and accurate documents. Thus, GDocP emerges as a non-negotiable element of any pharmaceutical quality system (PQS) and quality risk management (QRM).
What are Good Documentation Practices (GDocP)?
GDocP, also known as Good Recordkeeping Practice (GRK), is a set of protocols to ascertain informational validity and compliance with regulatory requirements in the pharmaceutical and medical device industries. GDocP supports a robust quality assurance system that is mandatory for organizations operating in these highly regulated sectors.
Different regional regulatory bodies, such as the FDA in the United States, the EMA in Europe, the WHO, Health Canada, and the TGA in Australia, have established comprehensive documentation requirements to adhere to Good Documentation Practices (GDocP). While some GDocP standards are codified by authorities, others are not but are considered part of cGMP, with an emphasis on “current.” These practices ensure that all records are precise, complete, timely, legible, traceable, and properly controlled.
Non-compliance with these mandates can lead to severe consequences, including regulatory penalties and fines, product recalls, and patient safety risks. Regulatory bodies may issue warning letters, suspend operations, or impose substantial fines. Additionally, poor documentation can compromise product quality, resulting in recalls and patient harm, while non-compliance can damage a company’s reputation, cause loss of licenses, and lead to fraud charges. This highlights the critical importance of adhering to GDocP to protect patient safety and preserve the integrity of pharmaceutical products and medical devices.
Knowing the Key Principles of GDocP

The core principles of GDocP, known as the ALCOA+ principles, state that all documentation should be:
- Attributable – Each recorded task must be traceable to the individual or computerized system that performed it, including the time of the task. This requirement extends to any modifications to records, such as corrections, deletions, and changes, where it is essential to know who made the change, when it was made, and why.
- Legible – All records must be clear and readable to ensure they are understandable and useful. This applies to all information needed to be considered complete, including all original records or entries.
- Contemporaneous – Documentation of actions, events, or decisions should be made at the time they occur. This ensures the documentation accurately reflects what was done or decided and why, capturing the influences on decisions at that time.
- Original – The original record is the first capture of information, whether on paper (static) or electronically (dynamic, depending on system complexity). Information initially captured in a dynamic state should remain available in that state.
- Accurate – Records must truthfully represent facts. Ensuring accuracy involves several elements of a pharmaceutical quality system (PQS), including:
- Equipment-related factors such as qualification, calibration, maintenance, and computer system validation.
- Policies and procedures that control actions and behaviors.
- Deviation management, including root cause analysis, impact assessments, and corrective and preventive actions (CAPA).
- Trained and qualified personnel who understand the importance of following established procedures and documenting their actions and decisions.
- Complete – All critical information must be retained to fully understand an event, without loss or deletion. The level of detail required depends on the criticality of the information.
- Consistent – Information must be created, processed, and stored in a consistent manner through standardized policies and procedures, such as chronological sequencing, date formats, and units of measurement.
- Enduring – Records must be maintained in a way that preserves their integrity and accessibility throughout the entire retention period.
- Available – Records must be readily accessible in a readable format during the retention period for all authorized personnel, whether for routine release decisions, investigations, trending, audits, or regulatory inspections.
Applying these principles appropriately across all applicable GMP and GDP activities, together with other supporting elements of a pharmaceutical quality system, ensures the reliability of information used to make critical decisions regarding medicinal products.
Types of Documents Requiring GDocP Compliance in Pharma & Medical Device Industries
The manufacturing processes of pharmaceutical products and medical devices involve distinct procedures and corresponding documentation to ensure quality, traceability, and regulatory compliance.
Standard Operating Procedures (SOPs)
SOP documents outline the essential processes and protocols that must be followed to ensure consistent product quality and adherence to regulatory requirements.
Quality Management System (QMS) Documentation
QMS documentation provides a structured framework for managing quality policies, procedures, and processes across the organization.
Batch Records & Production Logs
These records require strict GDocP compliance to maintain full traceability and control of manufacturing processes and product history.
Laboratory Notebooks & Testing Records
These documents capture data and observations from laboratory experiments and testing, ensuring data integrity, authenticity, and reproducibility of results.
Deviation & CAPA Reports
These reports document deviations from approved procedures and the corrective and preventive actions implemented to address root causes.
Training Records
Training records track employee qualifications and training history, ensuring personnel are competent to perform assigned tasks within regulated environments.
How to Ideally Implement a GDocP System
Like every other GxP, establishing a GDocP system should involve well-defined steps that cover the entire document lifecycle.
1. Establish a Documentation Policy
Develop a clear GDocP policy outlining objectives, scope, and applicability to ensure consistent documentation across all processes.
2. Develop Standard Operating Procedures (SOPs)
Create and standardize SOPs for document creation, review, approval, distribution, and archiving using approved templates.
3. Document Creation and Review
Ensure all documents are accurate, complete, legible, and traceable, with proper recording of data and essential details such as dates and signatures.
4. Document Approval and Distribution
Establish a thorough review and approval process involving authorized personnel and control document distribution to ensure only current versions are in use.
5. Record Keeping and Archiving
Store records securely, define retention periods in compliance with regulatory requirements, and ensure easy accessibility for authorized personnel.
6. Regular Audits and Reviews
Conduct routine internal audits to verify GDocP compliance, identify gaps, and implement continuous improvements.
7. Handling Deviations and Corrections
Document, investigate, and correct deviations from approved procedures while preserving original entries and preventing recurrence.
8. Documentation Culture
Promote a culture that recognizes proper documentation as essential to manufacturing excellence, supported by strong leadership commitment.
9. Electronic Documentation Systems
Implement electronic documentation systems to streamline document control, enhance accessibility, and ensure data integrity and regulatory compliance.
10. Training and Awareness
Provide comprehensive GDocP training to all employees, with periodic refreshers to maintain awareness and compliance.
Looking ahead, GDocP trends increasingly favor digital documentation and electronic records to improve efficiency and strengthen compliance. The transition from paper-based systems represents a significant operational shift for regulated organizations.
However, GDocP implementation presents challenges such as data security concerns, system validation requirements, and integration with legacy infrastructure. Addressing these challenges is critical to unlocking the full potential of a seamless and compliant GDocP system.
Major Challenges Faced in GDocP Implementation & How Smart Food Safe’s Smart Docs Helps
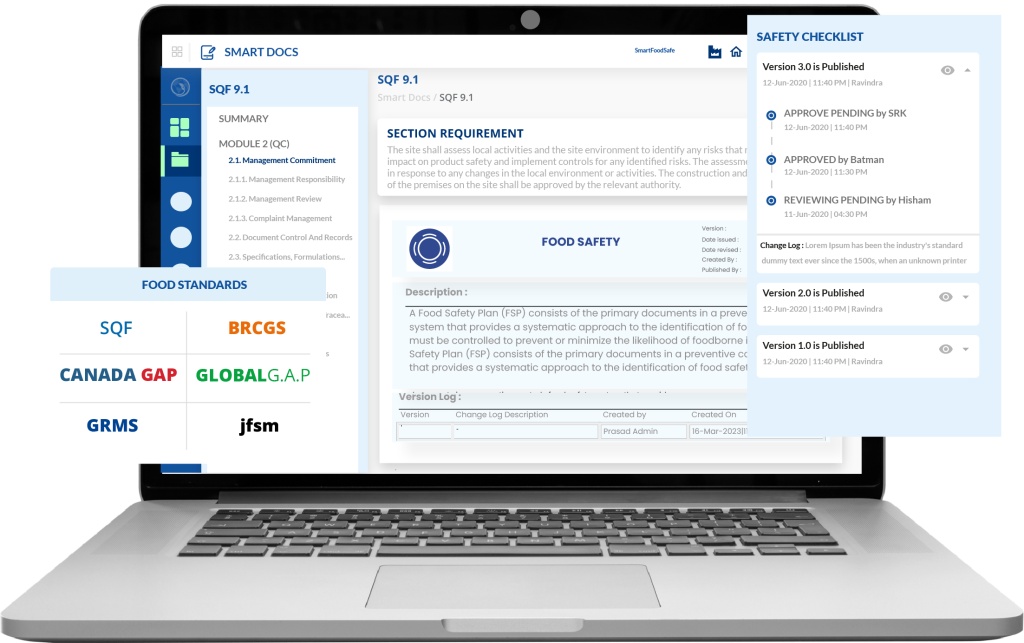
Smart Food Safe’s Smart Docs is a document compliance software solution designed to help businesses digitally implement and enforce Good Documentation Practices (GDocP).
It provides a centralized document management system that improves document control and transparency while addressing the key challenges that hinder effective GDocP implementation. Below, we explore how Smart Docs enables organizations to overcome these challenges.
| Challenges | How Smart Docs’ Features Assist |
|---|---|
|
Inconsistent Documentation Different personnel may document information in varied formats, leading to inconsistencies. |
Configuration of Custom Folders and Global Standards Properly categorize the documentation under uniform standards and folders. |
|
Human Error Mistakes in documentation, such as incorrect entries or miscalculations, are common and can affect data accuracy. |
Systematic Review and Approval Checks Well-defined workflow and reviewer notes to refer for respective users while approving the document, before submission for approval, and before publishing. |
|
Version Control Issues Maintaining the correct version of documents and ensuring that all personnel are using the most up-to-date version can be challenging. |
Version and Change Log Maintain the version and change log logic for each published document edit to track all changes and preserve document history. |
|
Retrieval and Accessibility of Documents Efficiently storing and retrieving documents, especially in large volumes, can be challenging. |
Document Numbering System Implement digital document numbering that allows organized indexing and retrieval of documents. |
|
Collaboration and Review Processes Facilitating collaboration and review processes among multiple stakeholders. |
Real-Time Document Collaboration Enable coauthoring for editing, viewing, commenting, and creating notes in real time while streamlining review and approval. |
|
Audit Readiness Being prepared for audits and inspections with accurate and easily accessible documentation. |
Audit Log Maintenance Comprehensive audit trails and reporting for each document version to quickly compile documentation for audits. |
|
Storage and Backup Managing the storage and backup of large volumes of documentation. |
Cloud-based Document Management Scalable storage solutions with automatic backups, eliminating the need for extensive physical storage. |
|
Document Integrity and Security Maintaining the integrity and security of documents to prevent unauthorized access or tampering. |
Access Controls and Encryption Ensure only authorized personnel can access and modify documents, maintaining integrity and security. |